
The alchemist’s ancient dream of turning one element into another had been banished once and for all. The second was that these atoms were stable and permanent. 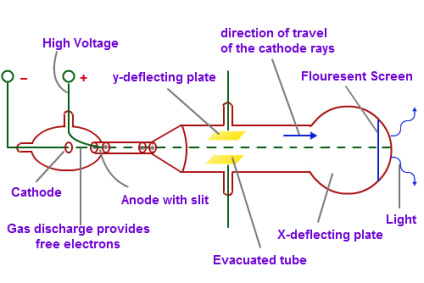
Indeed, the very word atom comes from the Greek atomos, meaning “uncuttable.” 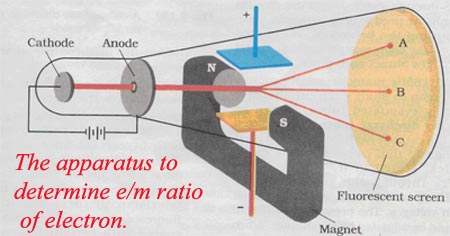
And most chemists believed there was no way to cut things any finer. Each element in the Periodic Table had its own unique kind of atom – that’s what distinguished one element from another. The first was that atoms were the smallest units of matter.Up to that point, nearly all scientists subscribed to two unshakeable beliefs about matter: One of the most profound shifts in our understanding of matter occurred over a 40-year period that began on the cusp of the 20th century. Ironically, in one sense the discovery of atomic structure proved the ancient Greeks correct: Matter really is made up of just a few things in combination – not air, water, earth and fire, but protons, neutrons and electrons. For this he won the Nobel Prize in 1938.įour German scientists, including longtime collaborators Otto Hahn and Lise Meitner, discovered that Fermi had actually split the uranium in half, into much lighter elements – an outcome almost no one had thought possible. Using neutrons to bombard uranium atoms, Italian physicist Enrico Fermi discovered what he thought were the first elements beyond uranium. The discoveries of Moseley and Rutherford raised a puzzling question about the atom: Why did the atomic weights of the elements rise so much faster than their atomic numbers? The puzzle was solved by James Chadwick’s 1932 discovery of the neutron. Darwin (grandson of the great biologist), and James Chadwick, who would win a Nobel Prize for his discovery of the neutron. Along with Harry Moseley, they included Hans Geiger (inventor of the Geiger counter), Charles G. Rutherford’s Manchester physics laboratory included one of the finest groups of scientists ever assembled in one place. 
This led to the discovery of the positively charged particle called the proton – and the realization that each element is defined by the number of protons in its nucleus: its atomic number. In 1913, physicist Harry Moseley discovered there was a simple relationship between an element’s X-ray spectrum and its atomic number. Rutherford’s vision of the atom: a dense core called the nucleus containing the atom’s positive charge and most of its mass, surrounded by tiny, negatively charged electrons, orbiting at a much greater distance from the nucleus than this drawing suggests. In 1910, Rutherford and two of his graduate students used radioactive alpha particles to make an astonishing discovery about the structure of the atom. That meant there must be smaller pieces inside, still awaiting discovery. Marie and Pierre Curie’s discovery that radium glows in the dark led to the realization that radioactivity was atoms falling apart and spitting out energy. Marie Curie’s study of the curious rays emanating from uranium led to the discovery of radioactivity and two new elements: polonium and radium. Thomson found that even when he used different metals for the cathode, the resulting electrons were always the same – a sign that the electron was a piece of every atom. An electrical current gave off a glow as it passed from the cathode at one end to the anode at the other. Thomson’s discovery of the electron came from his study of the Crookes tube, an early version of the cathode ray tubes that were later common in television sets. Thomson of Cambridge University discovered the electron in 1897, setting off a race to find the rest of the atom’s pieces. By the time physicists (including Marie Curie, Ernest Rutherford and Albert Einstein) gathered for the 1911 Solvay Conference, they had a pressing new question to answer: What’s inside the atom? CLICK TO ENLARGE
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
